Mastering Issue Resolution: A Guide to Corrective Actions

Streamlining Non-Conformities & Corrective Actions in ISO 9001 with AI
Non‑conformities are departures from defined requirements, and corrective actions are the structured steps taken to remove root causes and stop recurrence. Together they form the operational core of an effective ISO 9001 quality management system. This guide breaks down what non‑conformities look like, how to classify and document them, and how a disciplined corrective action process — driven by root cause analysis and modern AI‑assisted auditing — speeds resolution and reduces repeat issues. You’ll get a practical six‑step CAPA framework, a comparison of RCA techniques, clear guidance for meeting ISO 9001 Clause 10.2, and a look at how AI improves detection, prioritization, and verification. We also include checklists, EAV‑style comparison tables, and recommended next steps to help organizations tighten non‑conformity management and stay audit‑ready.
What Are Non‑Conformities in ISO 9001 and Why They Matter?
Non‑conformities occur when a product, process, or system doesn’t meet a specified requirement. They’re important because they expose weaknesses in the quality management system (QMS) that can harm performance, compliance, and customer confidence. Typically, an audit observation, customer complaint, or monitoring result uncovers the divergence and triggers documentation and corrective action. Fast, accurate classification and response reduce operational disruption and lower the chance of repeated failures — protecting reputation and cost. Knowing how to classify and measure impact helps you prioritize corrective work and allocate resources so the QMS stays aligned with business objectives and regulatory obligations.
How Are Different Types of Non‑Conformities Defined and Classified?
Auditors usually sort non‑conformities by severity and scope into categories like minor and major. A minor finding often points to an isolated procedural lapse (for example, a missing signature); a major finding indicates a systemic issue that affects product or service conformity, such as repeated process deviations. ISO 9001 itself doesn’t formally define a “critical” category, though some organizations or certifiers use that label informally for risks that threaten safety, data security, or legal compliance and require immediate action. Common decision criteria for classification are recurrence, scope, and severity — examples and thresholds help auditors decide when to escalate. Correct classification lets you target corrective actions and gather the right verification evidence for audit acceptance.
What Business and Compliance Risks Come from Unmanaged Non‑Conformities?
Left unchecked, non‑conformities raise audit risk, drive up corrective costs, and erode stakeholder confidence by letting defects or non‑compliant practices persist. Operationally, unresolved issues can cut throughput, increase waste, and strain supplier relationships when bad outputs flow downstream. Compliance consequences include fines, suspension of certification, or contract breaches; reputational damage can reduce customer and institutional trust. Repeated failures add remediation expense and weaken competitive positioning. Promptly addressing findings preserves certification standing, protects revenue, and supports continuous improvement.
- The primary financial risk is higher remediation costs and lost productivity.
- The primary compliance risk is audit failure or suspension of certification from systemic issues.
- The primary reputational risk is loss of stakeholder trust, which can threaten contracts and acceptance of certification.
Prioritizing non‑conformities effectively reduces these risks and keeps the QMS performing. Clear risk categories also guide corrective priorities during audit planning and management review.
What Are the Key Steps in the Corrective Action Process for ISO Compliance?
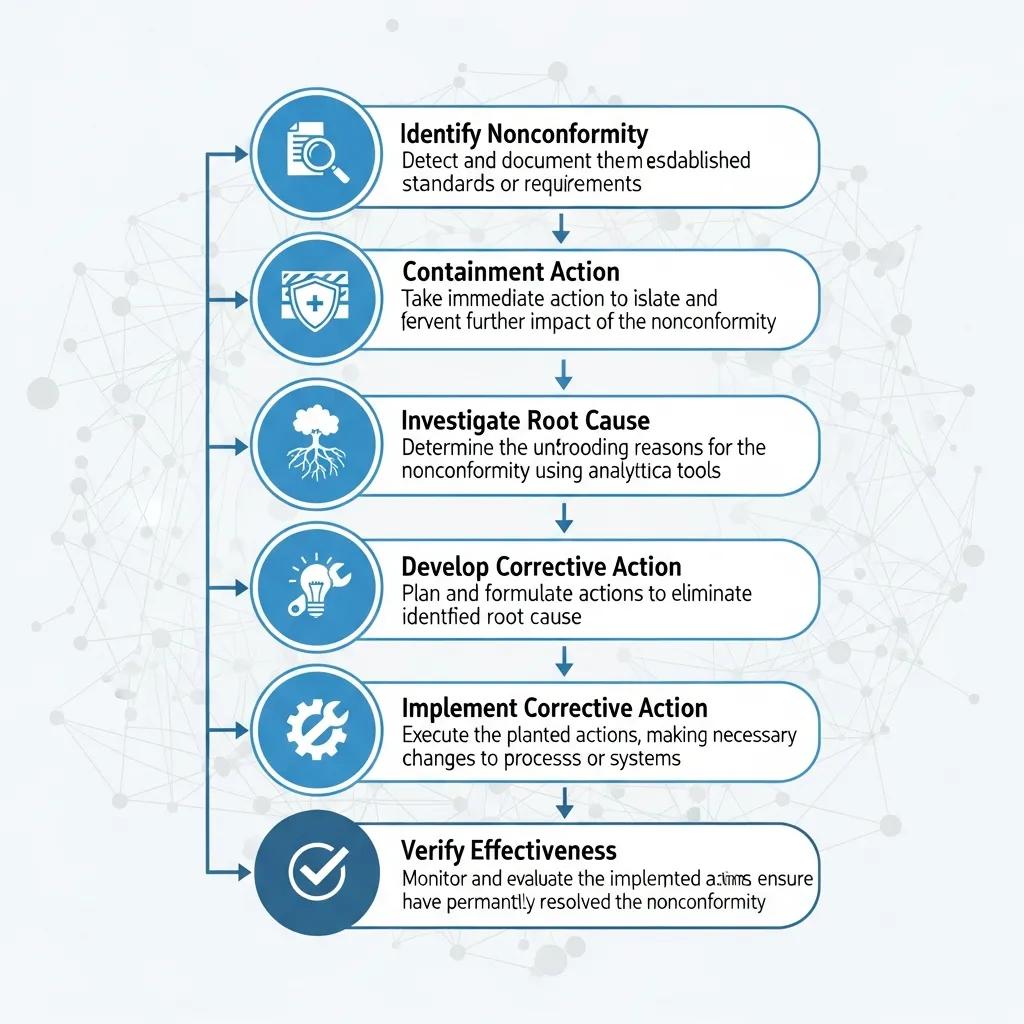
Corrective actions follow a lifecycle that runs from detection through closure and prevention. Each stage has a specific purpose and produces verifiable outputs for audit evidence: contain the problem, identify root causes, implement fixes, and confirm effectiveness. The clear outcome is a traceable record that satisfies ISO 9001 Clause 10.2 and supports continual improvement. Below is a practical six‑step framework plus an EAV‑style table mapping inputs, outputs, and accountable roles.
The way you detect and document a non‑conformity sets the tone for the whole corrective process — accurate reports make containment and root cause work far more effective.
- Identify and Record the Non‑Conformity: Capture the facts, affected outputs, and initial evidence.
- Containment and Interim Actions: Stop further non‑conforming outputs and protect customers or sensitive data.
- Root Cause Analysis (RCA): Use structured methods to find underlying causes.
- Corrective Action Plan: Specify actions, owners, deadlines, and resources.
- Implement Corrective Actions: Carry out fixes and update procedures and records.
- Verify Effectiveness and Close: Validate results, document evidence, and apply preventive measures.
These six steps create traceable verification evidence and meet audit closure expectations, showing both action taken and its effectiveness.
Each corrective phase calls for specific documentation standards so auditors can verify work during surveillance and recertification.
| Step | Purpose | Verification Example |
|---|---|---|
| Identification | Record what failed and where | Completed non‑conformity report with timestamped evidence |
| Containment | Prevent immediate recurrence or harm | Containment action record and affected batch hold logs |
| Root Cause Analysis | Find systemic reasons | RCA report (e.g., 5 Whys diagram) linked to data |
| Action Plan | Assign responsibility and resources | CAPA plan with owners and deadlines |
| Implementation | Execute and correct process inputs | Updated procedures and training records |
| Verification & Closure | Confirm effectiveness and prevent recurrence | Effectiveness review, metrics, and management sign‑off |
This mapping clarifies who does what and what evidence auditors expect at each corrective action phase, making responses consistent and verifiable.
How to Identify and Document Non‑Conformities Effectively?
Good identification pulls from multiple sources — internal audits, management reviews, customer complaints, monitoring, and incident reports — into concise, auditor‑ready records. Your documentation should include a clear description of the non‑conformity, objective evidence, affected products or processes, discovery date, and the reporter’s name to preserve traceability and avoid ambiguity. Examples of objective evidence are inspection records, test results, screenshots, or photos. Using structured templates with mandatory fields increases consistency and enables trend analysis, which helps prioritize corrective and preventive work across the QMS.
What Are the Six Essential Steps for Successful Corrective Actions?
The six‑step CAPA framework makes corrective actions systematic, measurable, and auditable. Each step reduces uncertainty: identification captures the issue, containment limits impact, RCA uncovers causes, planning assigns responsibility, implementation fixes the problem, and verification proves the fix worked. Where possible, verification should use quantitative criteria — defect‑rate reduction, zero recurrence over a set period, or restored control‑chart stability — so auditors have objective closure evidence. This lifecycle turns single incidents into organization‑level learning and prevention.
How Does Root Cause Analysis Support Non‑Conformity Resolution?
Root cause analysis shifts corrective action from symptom treatment to addressing systemic contributors, which reduces recurrence and strengthens the QMS. RCA is a structured inquiry that uncovers causal chains linking inputs, human factors, and environmental conditions to the failure. The result is more durable corrective measures and smarter preventive controls that lower risk and provide clearer audit evidence. When RCA outputs feed directly into CAPA plans, actions become proportionate and measurable, and a standardized RCA workflow helps scale lessons across sites and processes.
Which Root Cause Analysis Techniques Work Best for ISO Standards?
Choose RCA techniques to match problem complexity and available data. Use the 5 Whys for simple causal chains, Fishbone (Ishikawa) to visualize multi‑factor causes, Fault Tree Analysis for high‑risk technical failures, and Pareto analysis to prioritize recurring issues by frequency or impact. Each method has trade‑offs — speed versus depth, simplicity versus rigor — and combining techniques often delivers the clearest picture. Documenting the method chosen and why you used it demonstrates rigor to auditors and makes it easier to turn findings into corrective actions.
- 5 Whys: Fast and effective for single‑root causes.
- Fishbone: Visual categorization for multifactor problems.
- Fault Tree: Logical mapping for complex technical failures.
- Pareto: Prioritizes issues by frequency and impact.
Picking the right technique improves corrective quality and shortens verification cycles, making audits more predictable.
How to Embed Root Cause Analysis in the Corrective Action Process?
Make RCA a required phase in CAPA and define clear entry and exit criteria. Start with a documented problem statement, gather evidence, select an RCA method, and map causal factors to specific corrective actions with owners and deadlines. Link RCA outputs directly to the CAPA so every action addresses a named cause, and set verification metrics to measure effectiveness. Embedding templates and RCA training into the QMS ensures repeatability and builds organizational memory to prevent similar failures.
| RCA Phase | Task | Outcome |
|---|---|---|
| Problem Definition | Clarify symptoms and scope | Precise problem statement |
| Data Collection | Gather evidence and records | Evidence‑backed analysis |
| Method Selection | Choose 5 Whys, Fishbone, Fault Tree, etc. | Appropriate analytical lens |
| Analysis & Hypothesis | Identify root causes | RCA report with causal map |
| Action Mapping | Convert causes to actions | CAPA items with owners |
| Verification | Measure effectiveness | Closure evidence and metrics |
This mapping turns RCA from a theoretical exercise into practical work that drives verifiable corrective actions and continuous improvement.
How Does AI‑Assisted Auditing Improve Non‑Conformity Management?

AI‑assisted auditing strengthens non‑conformity workflows by automating detection, applying predictive risk scoring to prioritize issues, and speeding verification through evidence correlation. Techniques like anomaly detection on process data, natural language processing to scan reports and complaints, and pattern recognition to surface recurring issues help teams find problems faster and with more context. The practical payoff is higher detection rates, less manual work, and consistent prioritization that directs limited resources to the riskiest findings. AI also produces searchable audit trails and suggests verification checkpoints, helping improve closure rates and preparedness for surveillance audits.
What Benefits Does AI Bring to Detecting and Analyzing Non‑Conformities?
AI delivers measurable advantages: quicker anomaly discovery, identification of hidden patterns across datasets, and predictive signals that estimate recurrence risk before issues escalate. These capabilities reduce time‑to‑discovery, improve how you allocate corrective resources, and enable earlier interventions that lower downstream costs. AI can correlate inspection logs, maintenance records, and customer feedback to build richer case context and prioritize issues by modeled risk. Organizations that adopt AI‑supported auditing typically gain better trend visibility and more effective preventive planning.
- AI shortens detection time, lowering time‑to‑discovery for non‑conformities.
- AI uncovers cross‑dataset patterns that may be missed by manual review.
- AI supports predictive prioritization so teams focus on high‑risk issues first.
These benefits translate into faster closures and fewer repeat findings during audits.
| AI Feature | Function | Benefit & KPI Example |
|---|---|---|
| Anomaly Detection | Spot deviations in data streams | Faster discovery; KPI: 40% reduction in detection time |
| NLP Analysis | Extract themes from reports | Better root cause context; KPI: 30% more actionable findings |
| Predictive Scoring | Rank issues by recurrence risk | Prioritized response; KPI: 25% fewer repeat non‑conformities |
The table shows how specific AI features map to concrete audit and operational KPIs, improving non‑conformity management effectiveness.
How Does AI Help Automate Corrective Action Verification?
AI speeds verification by matching implemented actions to expected evidence patterns, checking record completeness, and flagging inconsistencies for human review. Workflows pair corrective actions with required evidence templates, then use automated checks to validate timestamps, signatures, updated procedures, and outcome metrics against predicted improvements. Typical KPIs include reduced verification time, fewer reopened CAPAs, and more consistent audit trails. Automated evidence matching builds a defensible record for auditors and management, raising confidence in closure decisions.
With the benefits clear, it’s useful to see how accredited providers bring these capabilities into certification workflows.
At Stratlane, we blend accredited ISO certification services with AI‑assisted auditing to speed detection and verification of non‑conformities while keeping evidence trails aligned with standards. As an accredited certification body operating across the US, EU, and UK, we integrate AI features — like anomaly detection and automated evidence correlation — into our audit services to improve closure rates and audit readiness. Our portfolio includes ISO Certification Services, Audit Services, and Certificate Management, and our auditors support organizations through certification and surveillance phases. If you’d like a quote or to schedule an audit, we can align AI‑enabled audit outputs with your certification and certificate‑management workflows.
What Do ISO Certification Requirements Say About Non‑Conformity and Corrective Actions?
ISO standards require organizations to address non‑conformities and take corrective actions proportionate to risk. ISO 9001 Clause 10.2 specifically asks for processes that respond to non‑conformities, control and correct them, and evaluate whether actions are needed to remove causes and prevent recurrence — including records and effectiveness reviews. Practically, this means a documented CAPA process, evidence of RCA, and verification of implemented measures. Meeting these requirements demonstrates continuous improvement and gives auditors the records they need. Mapping key clauses and evidence across ISO 9001, ISO 14001, ISO 27001, and ISO 42001 helps teams prepare for multi‑standard audits.
How Does ISO 9001 Clause 10.2 Define Non‑Conformity Management?
Clause 10.2 requires organizations to react to non‑conformities, take action to control and correct them, and evaluate the need to remove causes so issues don’t recur. It also requires records and review of action effectiveness. In practice, that means documenting non‑conformity reports, performing RCA, assigning corrective actions with clear ownership, and keeping verification and management‑review evidence. Auditors expect traceability from the initial finding through to closure and preventive steps, including data that show recurrence has been reduced. Turning Clause 10.2 into routine practice makes corrective work systematic and auditable.
| ISO Clause | Requirement Summary | Practical Evidence Example |
|---|---|---|
| ISO 9001 10.2 | React, correct, and prevent recurrence | Non‑conformity report, RCA, CAPA plan, verification records |
| ISO 14001 | Address environmental non‑conformities and impacts | Incident logs, corrective actions, environmental monitoring data |
| ISO 27001 | Manage information security incidents and corrective actions | Incident reports, control updates, access logs |
| ISO 42001 | Ensure AI governance non‑conformities are corrected | Governance action plans, algorithm change logs, validation reports |
How Do ISO 14001, ISO 27001, and ISO 42001 Affect Corrective Actions?
Each standard frames corrective actions within its risk domain. ISO 14001 focuses on environmental impacts and pollution prevention; ISO 27001 targets information security incidents and control remediation; ISO 42001 covers AI governance, transparency, and bias mitigation. Practical examples include remediating a pollution source under ISO 14001, patching and updating access controls under ISO 27001, and applying model governance updates under ISO 42001. Evidence varies by domain — environmental monitoring, security logs, and algorithm validation — but the corrective lifecycle (identify, analyze, act, verify) stays the same. That consistency lets integrated management systems use one CAPA process for multi‑standard compliance.
We support organizations across these standards by aligning certification workflows and certificate management with each standard’s corrective action expectations and by issuing certificates accepted by corporate and academic stakeholders. Our accredited auditors provide domain‑specific guidance and help map corrective evidence to clause requirements, assisting with surveillance and recertification planning.
How Can Businesses Leverage Stratlane’s AI‑Assisted ISO Certification Services?
Stratlane offers an integrated approach that pairs accredited ISO certification with AI‑assisted auditing to shorten time to detect non‑conformities and improve corrective verification. We use AI for anomaly detection and evidence correlation while our accredited auditors validate findings and ensure certificate issuance is accepted across the US, EU, and UK. Key differentiators are our experienced audit teams, AI tools that prioritize high‑risk findings, and certificate management services that track status and renewals. Clients gain streamlined audit workflows, clearer evidence trails for Clause 10.2 compliance, and support across ISO 9001, ISO 14001, ISO 27001, and ISO 42001 certification journeys.
What Makes Stratlane’s Approach to Non‑Conformity Management Different?
We combine AI‑driven detection with human auditor validation to balance automation speed with accreditation rigor. That hybrid model increases detection rates while preserving the expert judgment auditors require. Our accredited status and multi‑country certificate acceptance give organizations recognition across corporate and academic stakeholders. AI helps prioritize findings and automate evidence checks, while auditors focus on RCA quality and the robustness of corrective plans. The result is faster verification, less rework, and a stronger connection between corrective actions and continual improvement.
How Have Clients Benefited from Stratlane’s AI‑Enhanced Audit Process?
Clients tell us they’re more audit‑ready thanks to faster identification of systemic issues and shorter corrective cycles, leading to fewer repeat findings and cleaner evidence for surveillance audits. Typical, anonymized outcomes include shorter detection‑to‑closure timelines, more consistent CAPA records, and simplified certificate management during recertification. Our audit services, certificate management, and AI‑enabled verification free internal teams to focus on quality improvements rather than manual evidence collection. If you want tailored outcomes, request a quote or schedule an audit to estimate potential time and cost savings under our accredited, AI‑assisted certification model.
Frequently Asked Questions
What is the role of management in the corrective action process?
Management sets the tone and provides the resources required for effective corrective action. Leaders must prioritize quality, ensure staff understand reporting and corrective procedures, review corrective action reports, and allocate resources for implementation. Active management involvement creates accountability and aligns corrective work with organizational goals and ISO requirements.
How can organizations ensure continuous improvement in their corrective action processes?
Continuous improvement comes from regular review of non‑conformity data to spot trends, feeding lessons learned back into training and process updates, and running periodic audits and management reviews to test effectiveness. Establishing feedback loops, involving staff in quality initiatives, and keeping open channels for reporting help maintain momentum and drive ongoing improvement.
What are the common challenges faced during the corrective action process?
Common challenges include weak root cause analysis, low employee engagement, and poor documentation. Misdiagnosing causes leads to ineffective fixes; if staff aren’t empowered to report issues, problems go unaddressed; and inconsistent records make verification and traceability difficult. Addressing these gaps with training, clear templates, and leadership support reduces recurrence and improves outcomes.
How can technology improve the management of non‑conformities?
Technology streamlines data collection, analysis, and reporting. QMS software centralizes non‑conformity records, tracks corrective actions in real time, and provides analytics to spot trends. AI tools boost detection, prioritize risks, and suggest preventive measures based on historical data. Used well, technology increases efficiency, cuts manual errors, and strengthens ISO compliance.
What training is necessary for staff involved in the corrective action process?
Staff should be trained on applicable ISO standards, the organization’s QMS procedures, and how to identify and document non‑conformities. Training should include RCA techniques, clear communication skills, and any software tools used for CAPA. Regular refresher courses keep teams confident and aligned with evolving requirements.
How can organizations measure the effectiveness of their corrective actions?
Set clear KPIs: reduction in recurrence rates, average time to close issues, and number of repeat audit findings. Conduct follow‑up reviews to confirm sustained improvement and gather feedback from employees and stakeholders to understand perceived effectiveness. Use metrics plus qualitative insight to refine corrective strategies over time.
Conclusion
Managing non‑conformities and executing corrective actions is essential to maintain ISO 9001 compliance and improve quality performance. AI‑assisted auditing can accelerate detection, help prioritize risks, and simplify verification — all of which protect reputation and operational efficiency. This guide gives practical steps and tools to build a disciplined, auditable CAPA program that supports continuous improvement. To strengthen your QMS and prepare for certification, consider exploring Stratlane’s accredited ISO services and AI‑assisted audit offerings.